Ketamina E Depressione
#1

Inviato 09 dicembre 2011 - 10:40:57
Una ricerca dell'Università di Yale, diretta dal professor Ronald Duman e pubblicata su "Science"
Fattore K: la Ketamina contro i casi gravi di depressione
Il suo uso controllato per aiutare i pazienti resistenti alle normali cure farmacologiche
La Ketamina per combattere la depressione, specie quando si presenta in forme gravi o resistenti alle terapie convenzionali. E' l'idea proposta da una ricerca dell'Università di Yale, diretta dal professor Ronald Duman e pubblicata su "Science".
La Ketamina (detta anche Special K) è un composto usato come anestetico in diversi campi della medicina, dalia pediatria alla veterinaria.
Nella ricerca, gli studiosi hanno messo alla prova le sue facoltà, con test su cavie ed esseri umani.
Innanzitutto, la Ketamina è stata usata su topi con cellule celebrali danneggiate dallo stress cronico: il farmaco ha ripristinato i collegamenti sinaptici, aiutando la rigenerazione delle cellule lesionate.
Successivamente, è stata svolta una prova controllata con pazienti umani, sofferenti di depressione.
Allora, la Ketamina si è mostrata efficace nel 70% dei pazienti impermeabili alle normali terapie. Inoltre, "Il vantaggio è che una dose di farmaco può lavorare rapidamente per 7-10 giorni", spiega Duman.
Tuttavia è presto per dichiarare chiusa la pratica. A parte le cautele standard delle ricerche mediche, l'uso della Ketamina è di per sé insidioso. Come spiega il professore, la sostanza va somministrata per via endovenosa solo sotto stretto controllo medico, in quanto in grado di provocare sintomi psicotici a breve termine.
Fonte: Nanxin Li, Boyoung Lee, Rong-Jian Liu, Mounira Banasr, Jason M. Dwyer, Masaaki Iwata, Xiao-Yuan Li, George Aghajanian, and Ronald S. Duman, "mTOR-Dependent Synapse Formation Underlies the Rapid Antidepressant Effects of NMDA Antagonists", Science 20 August 2010 329: 959-964 [DOI: 10.1126/science.1190287] (in Reports)
Matteo Clerici
http://www.telegraph...depression.html
http://www.scienceda...00819141913.htm
--
http://www.politiche...-ketamina-.aspx
Il DPA sconsiglia utilizzo antidepressivo della ketamina
In riferimento alle recenti notizie sull'effetto antidepressivo della ketamina scoperto dal gruppo di ricercatori di Anita.E. Autry e pubblicato il 15 Giugno sulla rivista Nature il DPA precisa:
Affinché non vi siano malintesi e confusioni è utile chiarire molto bene che la sperimentazione a basse dosi di ketamina in pazienti affetti da depressione maggiore, rientra nella ricerca scientifica controllata per uso strettamente medico di tale sostanza. Va quindi precisato molto chiaramente che questo studio, del tutto preliminare, non può giustificare in alcun modo, ancorché auto -somministrato, l'uso di droga da strada a base di ketamina. Varie sostanze stupefacenti trovano un uso medico, ma va ricordato che le sostanze utilizzate per uso medico vengono prodotte con modalità estremamente controllate dalle aziende farmaceutiche e nonostante portino lo stesso nome di alcune droghe di uso comune, sono prodotti estremamente diversi (sia per la sicurezza che per l'efficacia) da quelle vendute in strada. Il DPA mette in guardia tutte le persone sofferenti di depressione a non utilizzare in alcun modo ketamina per tentare un' auto-cura assolutamente sconsigliata, non ammessa né opportuna per sintomi o condizioni quali la depressione. Allo stesso tempo, va ricordato il forte potenziale neurotossico della ketamina che è in grado di produrre gravi danni al sistema nervoso centrale e forti alterazioni neuropsichiche che possono mettere seriamente a rischio la vita dei pazienti se utilizzata fuori da un severo ed esclusivo controllo medico.
#2

Inviato 09 dicembre 2011 - 10:42:23
Uncovering the Biology of Depression
By Jonathan Hwang February 13, 2011
Professor of Psychiatry and Pharmacology Director at Yale University, Ronald Duman studies depression, a serious issue that affects approximately 17% of the U.S. population and is estimated to cost as much as $83.1 billion for our economy.
There is a general consensus in the scientific community that low levels of monoamine neurotransmitters are a major contributor of depression. From that belief, antidepressants are designed to increase levels of neurotransmitters in the brain. They block reuptake or breakdown of different types of neurotransmitters in order to maintain higher levels in the brain.
A diverse range of antidepressants exists. However, current antidepressants are still ineffective and have low response rates: only 1 in 3 respond to the first antidepressant treatment, and 2 in 3 ever respond after repeated treatments. Beyond knowing that antidepressants block or inhibit particular neurotransmitters, most scientists are still not sure which pathways these antidepressants influence and thus the types of mechanisms that contribute to depression. Dumans work investigates these unknown pathways.
Neurotrophic Theory of Depression
The bulk of Professor Dumans research has led to the formulation of the neurotrophic theory of depression, which states that neuronal growth factors contribute to the onset of depression. In 1995, Duman published a landmark paper relating increased brain derived neurotrophic factor (BDNF) levels with antidepressant effects, setting the foundation for the neurotrophic hypothesis of depression. Since then, subsequent research has clarified the pathways leading to the expression of BDNF. Professor Dumans research suggests that BDNF is expressed through the cyclic adenosine monophosphate (cAMP) signal transduction pathway, which is regulated by serotonin. In this pathway, cAMP is first activated, and this leads to activation of cAMP dependent protein kinase, which regulates cAMP response to element-binding protein (CREB). Additionally, Duman found that long-term antidepressants increase levels of CREB mRNA and protein in the hippocampus, further supporting the link between antidepressants and the cAMP pathway. CREB seems to regulate a set of genes in the hippocampus producing antidepressant effects.
The Role of BDNF
BDNF is a vital neurotrophic factor in the brain. Previous studies have shown that exposure to BDNF in the hippocampus can lead to increased strength in some synaptic connections. BDNFs role in neurogenesis was of particular interest. Duman discovered that upregulation of neurogenesis was the result of several antidepressants, suggesting that antidepressants reverse the atrophy of neurons that occur during depression. Other studies confirming Dr. Dumans work have found that increasing levels of BDNF in specific areas of the brain, such as the hippocampus, leads to antidepressant effects. The hippocampus has been implicated in mood disorders and its connections to amygdala and the prefrontal cortex are important for the function of cognition and emotion. Additionally, studies by Duman also revealed the converse: loss of BDNF contributes to depression. Stress, a precursor of many mood disorders, also decreases expression of BDNF.
The approaches Professor Duman took to clarify BDNFs role were varied. One was by using a viral vector to overexpress BDNF, which produced a behavioral phenotype typical of antidepressants. Antidepressant behavior was tested using the forced swim tests and learned helplessness models. Another approach involved infusing recombinant BDNF into the brain region, which also produced a similar antidepressant behavioral response. Duman also tested mice with a heterozygous deletion of the allele for BDNF. Although their phenotype appeared normal, they displayed a depressant-like phenotype once exposed to stress. This follows the widely held belief that a combination of environmental and genetic factors contributes to the onset of depression.
A Faster and More Efficient Pathway
In August 2010, Dumans lab discovered a completely new pathway, a major breakthrough for the field of depression. In ketamine, Duman addresses a pressing need in the field for a more rapid, more efficient drug to treat depression. In his paper published in Science, Duman lays the foundation for further understanding of this novel pathway.
Ten years ago, ketamine was preliminarily tested at the Connecticut Mental Health Center as an antidepressant in low doses. The subjects were patients who previously resisted all other forms of treatment, but over two thirds responded positively to ketamine. These results were confirmed in later studies. Much more remarkable about ketamines use as an antidepressant was how quickly the patients responded; antidepressant effects took place within two hours of treatment and lasted more than seven days.
The story is in the pathway, Duman explained. Unlike traditional antidepressants, which are generally neurotransmitter inhibitors, ketamine is a N-methyl-D-aspartic acid (NMDA) receptor antagonist. It operates on an entirely different pathway from those of traditional neurotransmitters. Studies conducted by graduate student Nick Li demonstrate that ketamine activates the mammalian target of rapamycin (mTor) pathway. mTor is a ubiquitous protein kinase involved in protein synthesis and synaptic plasticity in a process called synaptogenesis. Synaptogenesis restores the synapse connections in the brain that may deteriorate under stress and depression. The study also found increases in the levels of synapse proteins usually regulated by the mTor pathway. To physically confirm synapse formation, in collaboration with George Aghajanian, Professor of Psychiatry at Yale, two-photon imaging was used to observe increased spine density. Further supporting the link between the mTor pathway and antidepressant effects, Duman blocked the mTor pathway with rapamycin, leading to inhibition of ketamines antidepressant effects.
Ketamine is such a magic drug because it produces antidepressant effects in people who have resisted most other forms of treatment and its speed of response acts in days rather than weeks. However, the key disadvantages of directly using ketamine as an antidepressant are its use as a street drug and its toxicity from repeated dosages. Despite these shortcomings, knowing the mechanisms of ketamines antidepressant effects will further benefit drug designs for immediate antidepressant effects.
Dumans lab continues to further investigate factors and pathways for depression. One future direction is to deepen understanding of what stress does on a molecular scale and its link to depression. Other directions include studies to reveal more details about the mechanisms underlying Dumans neurotrophic theory of depression. Professor Dumans discoveries today could be the basis of depression treatments tomorrow.
About the Author
JONATHAN HWANG is a Chemical Engineering major in Saybrook College.
Acknowledgements
The author would like to thank Professor Ronald Duman for his time and assistance over the course of writing this article.
#3

Inviato 09 dicembre 2011 - 10:43:01
Ketamine Found To Act Like A Magic Drug On Depression
Thats according to the lead researcher of a team from Yale University in the US whose latest study suggests that ketamine, a drug normally used as an anesthetic, could be reformulated as an anti-depressant that takes effect in hours rather than the usual weeks and months of most available medications.
You can read how the researchers discovered this effect in a study they performed on rats which was published online on 20 August in the journal Science. Senior author Dr Ronald Duman, professor of psychiatry and pharmacology at Yale, told the media that just one dose of the drug can work rapidly and lasts for seven to ten days. This is the same ketamine that is used as a recreational drug, called Special K, or K.
He and his team found that the drug not only improved the rats depression-like behaviours, it also restored connections between neurons or brain cells that had been damaged by chronic stress. They called this synaptogenesis.
They hope their findings will help to speed up the development of a safe and easy to administer version of ketamine, which has already proved to be effective in severely depressed patients, they said. About ten years ago, scientists at Connecticut Mental Health Center found that in lower doses, ketamine, normally used as a general anesthetic for children, appeared to relieve patients with depression. Since then, other studies have shown that over two thirds of patients who dont respond to all other types of anti-depressants improved hours after receiving ketamine, said Duman.
The problem with using ketamine more widely to treat depression has been the fact it has to be given intravenously under medical supervision, and it can also cause short-term psychotic symptoms. So Duman and colleagues decided to investigate the effect of ketamine on the brain to see if it might reveal suitable targets for other safer and easier to administer drugs.
They found that ketamine acts on a pathway that controls the formation of new synaptic links between neurons, encouraging synaptogenesis; they wrote that they observed: increased synaptic signaling proteins and increased number and function of new spine synapses in the prefrontal cortex of rats.
Moreover, they found that a critical point on the pathway, involving the enzyme mTOR, controls production of proteins needed to form the new synapses. The researchers concluded that: Our results demonstrate that these effects of ketamine are opposite to the synaptic deficits that result from exposure to stress and could contribute to the fast antidepressant actions of ketamine.
Duman and colleagues told the press that they can already see ways to sustain the rapid effect of ketamine by intervening at other points downstream of this critical one. These could be additional targets for new drugs. This discovery not only brings new hope to the 40 per cent or so of patients with depression who dont respond to medication, but to many others who only experience relief after months and sometimes years of treatment.
The researchers also noted that ketamine has already shown to be effective as a rapid way to treat people with suicidal thoughts, many such patients usually only respond weeks later with traditional drugs.
Catharine Paddock, PhD, Medical News Today
#4

Inviato 09 dicembre 2011 - 10:44:06
Ketamine and Major Depressive Disorder: Is it Better with Special K?
By Scicurious | July 14, 2011
Most people have heard of ketamine. Originally invented in 1962 to be used as an anesthetic, it is still used for children and in some topical anesthetics, but mostly when you hear of ketamine used clinically now, its actually used in combination with xylazine as a veterinary anesthetic (side note: SciCat coming to after a visit to the vet from a Ketamine/Xylazine combo is hilarious. Hilarious and full of ANGER).
But of course, the medical uses of ketamine are not what people have heard about. Instead, people hear about the recreational uses of ketamine (aka Special K), where street users describe hallucinations and a sense of dissociation from the world. Its achieved widespread fame as a drug of abuse, and thats how most people know it nowadays.
But there may be more to it than that. There are currently trials underway to look at how ketamine treatment might help with depression and other psychiatric disorders in humans.
Of course, you can do clinical trials in humans and get subjective reports from the patients. But if you want to see whats REALLY going down with how ketamine is WORKING, you need a brain. And for brains, you need rats.
Li et al. Glutamate N-methyl-D-aspartate receptor antagonists rapidly reverse behavioral and synaptic deficits caused by chronic stress exposure Biological Psychiatry, 2011.
Ketamine does not behave like many other drugs that are known to be drugs of abuse. Unlike the benzodiazepines, it doesnt act on GABA. It doesnt act directly on dopamine like the stimulants, or on serotonin like the hallucinogens. Instead, ketamine acts on the neurotransmitter glutamate, the main excitatory neurotransmitter in the brain. But not DIRECTLY. Instead, it acts on a specific type of glutamate RECEPTOR, the N-methyl-D-aspartate (NMDA) receptor type. Ketamine acts at NMDA receptors as an antagonist, blocking the ability of glutamate to bind to the receptor and do its job.
And this is an aspect of the brain that has been relatively ignored in studies of depression. Often, scientists who study depression focus on serotonin, and on the birth of new neurons in the brain. Many believe that the increases in serotonin produced by traditional antidepressants such as Prozac lead to the increases in neurogenesis in the hippocampus which may help symptoms of depression. But serotonin doesnt have to be the ONLY neurotransmitter involved. There could be other mechanisms that mediate how depression occurs, and thus other potential drug targets.
And we NEED some other drug targets. Major Depressive Disorder is widespread, and in a large number of cases, the available antidepressants never work. So recently scientists have begun to look at glutamate, and at ketamine. Clinical trials have shown that low doses of ketamine (which still can produce some dissociative symptoms, but no hallucinations) can produce a rapid antidepressant response in severely depressed or bipolar patients. The studies are still very small and limited. And so far, its unknown HOW ketamine is acting to relieve depression in these patients.
Bring on the rats.
For this study, the authors took rats, and induced a depression-like state (we say depression-like because you cant ever ASK a rat how it feels about life) using a method called Chronic Unpredictable Stress. Kind of like exposing a rat to the equivalent of grad school. The stresses could be anything, and the rats get two stressors per day. But instead of experimental equipment breaking or their advisor yelling at them or running out of beer money, the rats get stressors like being placed in a chilly room, leaving the lights on overnight, bad smells, being put on a shaker plate or having their cages tilted at weird angles, or leaving the radio on loud. After 21 days of this, you get some stressed out unhappy rats. You can tell by giving them a test to see how much sugar water they want to drink. Happy rats LOVE sugar water, but unhappy rats will drink less of the sugar water.
Here you can see the results for the sucrose drinking rats exposed to chronic stress. The rats showed a decrease in sucrose preference, as well an increase in how long it took them to eat food in a novel environment (called Novelty Suppressed Feeding). BUT, when they gave the rats a single dose of either ketamine or a similar NMDA antagonist RO-256981, 24 hours before they began testing, the rats didnt have these symptoms. They drank as much sucrose water and ate as much food as animals that had never been exposed to stress at all. And in the bottom two panels of the figure you can see that this effect of a single dose of ketamine or RU-256981 lasted for up to 7 days after the drug was given to the stressed animals.
But of course, if you want to determine a mechanism of how something is acting in the body, you have to BLOCK it. In this case, the authors gave a drug called rapamycin, which is a bacterial produce that inhibits the mammalian target of rapamycin, otherwise called mTOR (you know that you know NOTHING about a protein when you call it oh, you know, that target of that one drug we have ). Luckily, we do NOW know a good bit about mTOR, which is a kinase that regulates things like cell growth and proliferation, as well as transcription of DNA. IT also lies downstream of NMDA receptor signaling, so is probably stimulated by drugs which hit the NMDA receptor. So IF ketamine is relieving anhedonia in these rats via mTOR, blocking mTOR will block the effects of ketamine.
So they gave rapamycin right before giving ketamine in the stressed out rats, and rapamycin blocked the effects of ketamine on sucrose preference and suppressed feeding. The rats looked as stressed as ever when mTOR was blocked, which suggests that ketamine was producing the behavioral effects via mTOR.
The authors then looked for various proteins that could be involved. They found that proteins that are associated with synapses, like glutamate receptors and proteins like synapsin 1 are reduced during stress in the rats, and that ketamine can increase these proteins again.
But what are these proteins DOING? It looks like they may be involved in difference spine densities in the stressed rat brains. They authors looked at neurons in the prefrontal cortex, looking specifically at an area called the apical tuft, which is where the tuft of dendrites comes out at the end of the axon (more on basic neuron anatomy here). This is because depressive symptoms in animals are associated with something called dendritic atrophy, where you get a decrease in the numbers of dendritic spines in areas like the apical tuft.
You can see here the photos of the dendritic spines from the rat prefrontal cortex (yup, we can take pictures of tiny parts of tiny neurons. Sometimes, that STILL blows my mind). The stressed rats have decreases in the number of little spines coming off the dendrites, and this can be reversed with ketamine.
But finally, we want to know how stress, and then ketamine, changes the way the neurons BEHAVE. To figure that out, we have to do electrophysiology, which is a technique where you take a REEEAAAALLY TEEEENY end of a glass tip, and suck a REAAAALLLLLY TEEEENY bit of cell membrane into it. If you do this in a live brain slice (which you can keep alive for a few hours outside of the rats head), you can get a live cell, and youve got something patched into it. You can then get recordings of what the cell is doing electrically (how it is firing, action potentials, etc), kind of like using peephole into a room.
In this case, they were interested two specific TYPES of neurons, those receiving the neurotransmitter serotonin, and those received the relatively new transmitter hypocretin/orexin (so new they are still arguing about the name). Those receiving serotonin are involved in signaling within the cortex, while those receiving hypocretin are involved in signaling which goes outside the cortex to the thalamus.
So they put serotonin and hypocretin on their slices and looked at how the neurons behaved.
You can see the little traces there, which show the neurons experiencing little postsynaptic currents. In the stressed rats (center) the currents were reduced in both cases, but when you gave ketamine, they were increased again. This may mean that the lack of dendritic spines seen in the stressed animals has functional effects on how well the neurons can make their little postsynaptic currents, which is a big effect on functionality.
What I find to be most interesting about this study is that there was only ONE DOSE of ketamine given here. ONE. The effects lasted up to a week. We dont know if wed get similar effects in humans (or whether the rats were experiencing hallucinations, for that matter, tough to ask them about that), but if we did, its possible that the mechanism through which ketamine works could be used to find new and effective antidepressant drugs. Or, if the effects of ketamine are mostly temporary and its not feasible to give it as a long term drug (and it may not be due to legal issues and the potential for abuse and thus possibly the selling of it to other people), we may be able to give it in the clinic, and use it to kickstart the effects of more traditional antidepressants like Prozac, where the ketamine may be able to bridge the gap while the Prozac is working (though no results yet on whether the ketamine increases neurogenesis like other antidepressants do, but knowing the work of this laboratory, I bet theyre on it). Or maybe well get both. I dont know if its a magic bullet (I doubt it), but I think its got potential as a new mechanism to pursue when looking for antidepressant drugs.
#5

Inviato 09 dicembre 2011 - 10:45:47
http://www.ncbi.nlm....pubmed/20724638
Science. 2010 Aug 20
mTOR-dependent synapse formation underlies the rapid antidepressant effects of NMDA antagonists.
Li N, Lee B, Liu RJ, Banasr M, Dwyer JM, Iwata M, Li XY, Aghajanian G, Duman RS.
Source
Laboratory of Molecular Psychiatry, Center for Genes and Behavior, Department of Psychiatry, Yale University School of Medicine, 34 Park Street, New Haven, CT 06508, USA.
Abstract
The rapid antidepressant response after ketamine administration in treatment-resistant depressed patients suggests a possible new approach for treating mood disorders compared to the weeks or months required for standard medications. However, the mechanisms underlying this action of ketamine [a glutamate N-methyl-D-aspartic acid (NMDA) receptor antagonist] have not been identified. We observed that ketamine rapidly activated the mammalian target of rapamycin (mTOR) pathway, leading to increased synaptic signaling proteins and increased number and function of new spine synapses in the prefrontal cortex of rats. Moreover, blockade of mTOR signaling completely blocked ketamine induction of synaptogenesis and behavioral responses in models of depression. Our results demonstrate that these effects of ketamine are opposite to the synaptic deficits that result from exposure to stress and could contribute to the fast antidepressant actions of ketamine.
-
http://www.ncbi.nlm....pubmed/21907221
2011 Sep 2.
Signaling pathways underlying the rapid antidepressant actions of ketamine.
Duman RS, Li N, Liu RJ, Duric V, Aghajanian G.
Source
Departments of Psychiatry and Neurobiology, Yale University School of Medicine, 34 Park Street, New Haven, CT 06508, USA. ronald.duman@yale.edu
Abstract
Currently available medications have significant limitations, most notably low response rate and time lag for treatment response. Recent clinical studies have demonstrated that ketamine, an NMDA receptor antagonist produces a rapid antidepressant response (within hours) and is effective in treatment resistant depressed patients. Molecular and cellular studies in rodent models demonstrate that ketamine rapidly increases synaptogenesis, including increased density and function of spine synapses, in the prefrontal cortex (PFC). Ketamine also produces rapid antidepressant actions in behavioral models of depression, and reverses the deficits in synapse number and behavior resulting from chronic stress exposure. These effects of ketamine are accompanied by stimulation of the mammalian target of rapamycin (mTOR), and increased levels of synaptic proteins. Together these studies indicate that ketamine rapidly reverses the atrophy of spines in the PFC and thereby causes a functional reconnection of neurons that underlies the rapid behavioral responses. These findings identify new targets for rapid acting antidepressants that are safer than ketamine. This article is part of a Special Issue entitled 'Anxiety and Depression'.
-
http://www.ncbi.nlm....pubmed/21996102
2011 Oct 11.
Cell atrophy and loss in depression: reversal by antidepressant treatment.
Banasr M, Dwyer JM, Duman RS.
Source
Departments of Psychiatry and Neurobiology, Yale University School of Medicine, 34 Park Street, New Haven, CT 06508, United States.
Abstract
Depression is associated with structural alterations in limbic brain regions that control emotion and mood. Studies of chronic stress in animal models and postmortem tissue from depressed subjects demonstrate that these structural alterations result from atrophy and loss of neurons and glial cells. These findings indicate that depression and stress-related mood disorders can be considered mild neurodegenerative disorders. Importantly, there is evidence that these structural alterations can be blocked or even reversed by elimination of stress and by antidepressant treatments. A major focus of current investigations is to characterize the molecular signaling pathways and factors that underlie these effects of stress, depression, and antidepressant treatment. Recent advances in this research area are discussed and potential novel targets for antidepressant development are highlighted.
-
http://www.ncbi.nlm....pubmed/22036038
2011 Oct 28.
Brain-Derived Neurotrophic Factor Val66Met Allele Impairs Basal and Ketamine-Stimulated Synaptogenesis in Prefrontal Cortex.
Liu RJ, Lee FS, Li XY, Bambico F, Duman RS, Aghajanian GK.
Source
Department of Psychiatry, Yale School of Medicine, New Haven, Connecticut.
Abstract
BACKGROUND:
Knock-in mice with the common human brain-derived neurotrophic factor (BDNF) Val66Met polymorphism have impaired trafficking of BDNF messenger RNA to dendrites. It was hypothesized, given evidence that local synapse formation is dependent on dendritic translation of BDNF messenger RNA, that loss-of-function Met allele mice would show synaptic deficits both at baseline and in response to ketamine, an N-methyl-D-aspartate antagonist that stimulates synaptogenesis in prefrontal cortex (PFC).
METHODS:
Whole-cell recordings from layer V medial PFC pyramidal cells in brain slices were combined with two-photon laser scanning for analysis of wildtype, Val/Met, and Met/Met mice both at baseline and in response to a low dose of ketamine.
RESULTS:
Val/Met and Met/Met mice were found to have constitutive atrophy of distal apical dendrites and decrements in apically targeted excitatory postsynaptic currents in layer V pyramidal cells of PFC. In addition, spine density and diameter were decreased, indicative of impaired synaptic formation/maturation (synaptogenesis). In Met/Met mice the synaptogenic effect of ketamine was markedly impaired, consistent with the idea that synaptogenesis is dependent on dendritic translation/release of BDNF. In parallel behavioral studies, we found that the antidepressant response to ketamine in the forced swim test was blocked in Met/Met mice.
CONCLUSIONS:
The results demonstrate that expression of the BDNF Met allele in mice results in basal synaptic deficits and blocks synaptogenic and antidepressant actions of ketamine in PFC, suggesting that the therapeutic response to this drug might be attenuated or blocked in depressed patients who carry the loss of function Met allele.
-
http://www.ncbi.nlm....pubmed/22114864
2011 Nov 24
mTOR activation is required for the antidepressant effects of mGluR2/3 blockade.
Dwyer JM, Lepack AE, Duman RS.
Source
Yale University School of Medicine, New Haven, CT, USA.
Abstract
Recent studies demonstrate that ketamine, a fast-acting antidepressant, rapidly activates the mammalian target of rapamycin (mTOR) and increases synaptogenesis in the prefrontal cortex. Because of the side-effect and abuse potential of ketamine we are investigating alternative agents that produce similar effects. Here, we demonstrate that a single dose of LY 341495, an mGluR2/3 antagonist, produces ketamine-like biochemical and behavioural actions. LY 341495 administration rapidly (1 h) activates the mTOR pathway (mTOR, p70S6K, 4E-BP1) and subsequently (24 h later) increases levels of synaptic proteins (PSD-95, GluR1 and Synapsin I), similar to the effects of ketamine. Finally, the antidepressant effects of LY 341495 in the rat forced swim test are completely blocked by the mTOR inhibitor, rapamycin. The results indicate that the antidepressant actions of LY 341495 are mediated by activation of mTOR and suggest that this and other mGluR2/3 antagonists could produce rapid antidepressant effects in depressed patients.
#6

Inviato 09 dicembre 2011 - 10:46:54
Nature. 2011 Jun 15;475(7354):91-5. doi: 10.1038/nature10130.
NMDA receptor blockade at rest triggers rapid behavioural antidepressant responses.
Autry AE, Adachi M, Nosyreva E, Na ES, Los MF, Cheng PF, Kavalali ET, Monteggia LM.
Source
Department of Psychiatry, University of Texas Southwestern Medical Center, 5323 Harry Hines Boulevard, Dallas, Texas 75390-9111, USA.
Abstract
Clinical studies consistently demonstrate that a single sub-psychomimetic dose of ketamine, an ionotropic glutamatergic NMDAR (N-methyl-D-aspartate receptor) antagonist, produces fast-acting antidepressant responses in patients suffering from major depressive disorder, although the underlying mechanism is unclear. Depressed patients report the alleviation of major depressive disorder symptoms within two hours of a single, low-dose intravenous infusion of ketamine, with effects lasting up to two weeks, unlike traditional antidepressants (serotonin re-uptake inhibitors), which take weeks to reach efficacy. This delay is a major drawback to current therapies for major depressive disorder and faster-acting antidepressants are needed, particularly for suicide-risk patients. The ability of ketamine to produce rapidly acting, long-lasting antidepressant responses in depressed patients provides a unique opportunity to investigate underlying cellular mechanisms. Here we show that ketamine and other NMDAR antagonists produce fast-acting behavioural antidepressant-like effects in mouse models, and that these effects depend on the rapid synthesis of brain-derived neurotrophic factor. We find that the ketamine-mediated blockade of NMDAR at rest deactivates eukaryotic elongation factor 2 (eEF2) kinase (also called CaMKIII), resulting in reduced eEF2 phosphorylation and de-suppression of translation of brain-derived neurotrophic factor. Furthermore, we find that inhibitors of eEF2 kinase induce fast-acting behavioural antidepressant-like effects. Our findings indicate that the regulation of protein synthesis by spontaneous neurotransmission may serve as a viable therapeutic target for the development of fast-acting antidepressants.
#7

Inviato 09 dicembre 2011 - 11:03:26
2011 Jun 1 Epub 2011 Apr 3.
Rapid decrease in depressive symptoms with an N-methyl-d-aspartate antagonist in ECT-resistant major depression.
Ibrahim L, Diazgranados N, Luckenbaugh DA, Machado-Vieira R, Baumann J, Mallinger AG, Zarate CA Jr.
Source
Experimental Therapeutics & Pathophysiology Branch, Intramural Research Program, National Institute of Mental Health, National Institutes of Health, 10 Center Drive, CRC Unit 7 Southeast, Room 7-3445, Bethesda, MD 20892, USA.
Abstract
BACKGROUND:
Ketamine rapidly improves depressive symptoms in patients with treatment-resistant major depressive disorder (MDD) who do not respond to multiple standard antidepressants. However, it remains unknown whether ketamine is equally effective in patients with MDD who previously also did not respond to electroconvulsive therapy (ECT).
METHODS:
This study compared 17 patients with treatment-resistant MDD who previously did not respond to ECT and 23 patients with treatment-resistant MDD who had not previously received ECT. All subjects received a single open-label infusion of ketamine (0.5 mg/kg). Patients were evaluated using the Montgomery-Asberg Depression Rating Scale (MADRS) at baseline (60 min before the infusion), as well as at 40, 80, 120, and 230 min after infusion.
RESULTS:
Depressive symptoms were significantly improved in the ECT-resistant group at 230 minutes with a moderate effect size (p < .001, d = 0.50, 95% C.I.: 0.21-0.80). At 230 minutes, the non-ECT exposed group showed significant improvement with a large effect size (p < .001, d=1.00, 95% C.I.: 0.71-1.29).
CONCLUSION:
Ketamine appears to improve depressive symptoms in patients with MDD who had previously not responded to ECT. These preliminary results encourage further investigation with a larger sample size to determine effectiveness compared to other treatment-resistant patients with MDD.
#9

Inviato 09 dicembre 2011 - 15:23:17
non mi pare che dalla SPET, emerga un quadro di depressione, ma anzi...
Sniper?con chi parli..?chi ti ha detto che siamo depressi...almeno nella comune accezione del termine...No: esiste una depressione reattiva per la malattia invalidante che ci tortura; come è pur vero, che la parola "depressione" è usata impropriamente.
#10

Inviato 09 dicembre 2011 - 18:03:17
14. La lingua utilizzata in questo forum è l'italiano, fatta eccezione per l' "AREA INTERNAZIONALE", non sono dunque ammesse altre lingue, se non accompagnate da relativa, integrale traduzione in italiano e breve introduzione che presenti una sintesi del testo tradotto che verrà riportato di seguito.
#12

Inviato 13 aprile 2012 - 23:22:00
Messaggio modificato da romy, 13 aprile 2012 - 23:26:57
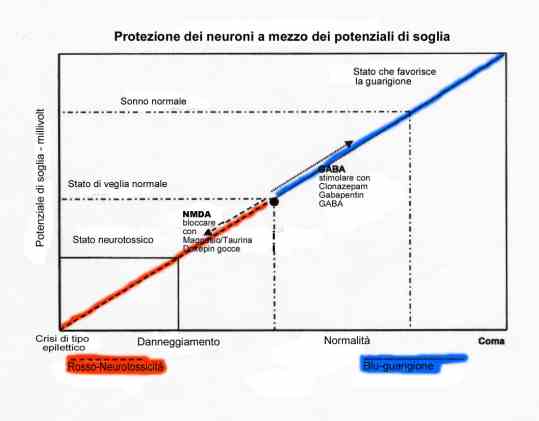
Iosto'conChiara
Quando le voci in te parlano di fine;
quando la mente dice che hai perduto;
quando credi che sia impossibile;
eppure prosegui,ti sollevi sulla tua Spada;
e fai ancora un altro passo;
Lì è dove termina l'Uomo;
Lì è dove comincia Dio.
Mentre si aspettano future ricerche è importante per prima cosa non
nuocere.
Non è vero che i giorni di pioggia sono i più brutti, sono gli unici in cui puoi camminare a testa alta anche se stai piangendo.
"Nessun Medico può dire che una malattia é Incurabile.
Affermarlo é come offendere Dio, la Natura e disprezzare il Creato.
Non esiste malattia, per quanto terribile possa essere,
per la quale Dio non abbia una cura corrispondente".
Paracelso
“Questa realtà è solo un sogno di cui siamo convinti!”
Mio collegamento
Per quanto riguarda l'ipotesi che la CFS possa essere una forma di Depressione Mentale,tutti gli studi hanno contraddetto tale approccio.Per citare un solo ma importante rilievo clinico:i livelli di Cortisolo sono molto bassi nella CFS,al contrario di quelli alterati verso l'alto della Sindrome Depressiva.
Allora, se capiamo che siamo responsabili di ciò che viviamo, già questo cambia del tutto la visione delle cose.
Livello anormalmente alto o basso di cAMP causa difetti di apprendimento e di capacità di memoria,in generale.
Sul cAMP ci sarebbero molte cose da dire al fine di una buona memoria e cognitività,forse lo faro' un giorno sul mio topic,tempo permettendo,ora voglio solo ripetere una verità a cui sono arrivato da qualche anno,aumentare il cAmp nei giovani comporta un miglior apprendimento e memoria,accade l'inverso nelle persone adulte o anziane.Quindi il cervello dei giovani si comporta all'opposto dei cervelli dei vecchi ed anziani questo avviene anche negli animali da laboratorio,questo spiega anche tutta la diatriba sulla Cannabis terapeutica..........negativa nei giovani un toccasana negli anziani,perchè comporta una diminuzione,appunto,del cAmp e quindi potenzia la comunicazione tra neuroni e quindi la memoria,apprendimento e cognitività,mal ridotti nella CFS/ME,fibromialgia,MCS,MBS,ed altre......
https://www.youtube....h?v=ICjFAa2ZbIY

Aggiungi risposta veloce
1 utente(i) stanno leggendo questa discussione
0 utenti, 1 ospiti, 0 utenti anonimi
Tutti i testi degli articoli e la veste grafica sono proprietà esclusiva di CFSItalia.it e ne è vietata la riproduzione, totale o parziale,
senza esplicita autorizzazione da parte degli amministratori.
Le informazioni fornite in questo Forum non intendono sostituirsi né implicitamente né esplicitamente al parere professionale di un medico né a diagnosi, trattamento o prevenzione di qualsivoglia malattia e/o disturbo. CFSItalia.it non ha alcun controllo sul contenuto di siti terzi ai quali sia collegato tramite link e non potrà essere ritenuto responsabile per qualsivoglia danno causato da quei contenuti. Gli amministratori e moderatori non sono responsabili dei messaggi scritti dagli utenti sul forum.







